|
Thus, the molar mass of bilirubin can be expressed as 584.05 g/mol, which is read as “five hundred eighty four point zero five grams per mole. The division sign (/) implies “per,” and “1” is implied in the denominator. We can rearrange the key equation and multiply moles by molar mass, substituting in the mole value that we have just calculated, 1,492.23 moles. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. 
The mass of 1 mol of bilirubin is 584.69 g.īe careful when counting atoms. Anyway the question goes onto to ask you to work out the mass of aluminium, I know that mass moles x molar mass and that 2Al and 2KOH are in a 2 : 2 moles. For example, take the example of zinc nitrate, or Zn (NO 3) 2. M is the molar mass of aluminum, I is the fractions, respectively. For any substance (element or compound), the molar mass is the mass of that substance in grams divided by its. The molar mass will be equal to: (1 atom x 56 grams/mole Fe) (2 atoms x 35.5 grams/mole of chlorine) 127 grams/mole of iron (II) chloride For other compounds, this might get a little bit more complicated. 3) atomic mass of aluminium is 27 g/mole. Walloy contraction of aluminum during solidification 5. 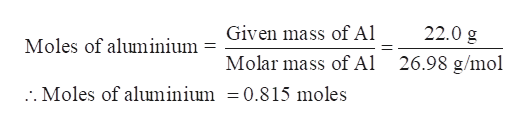
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
